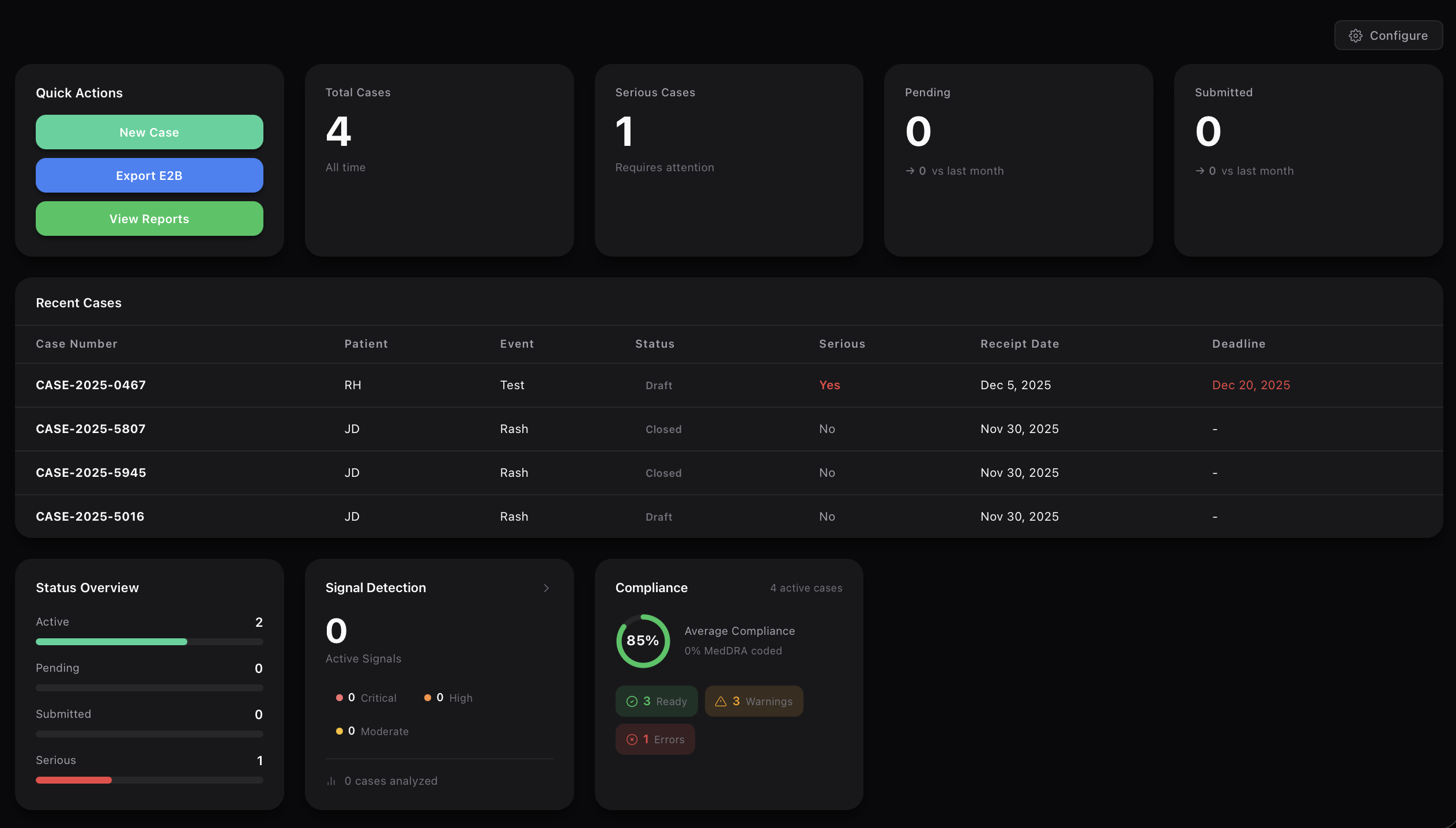


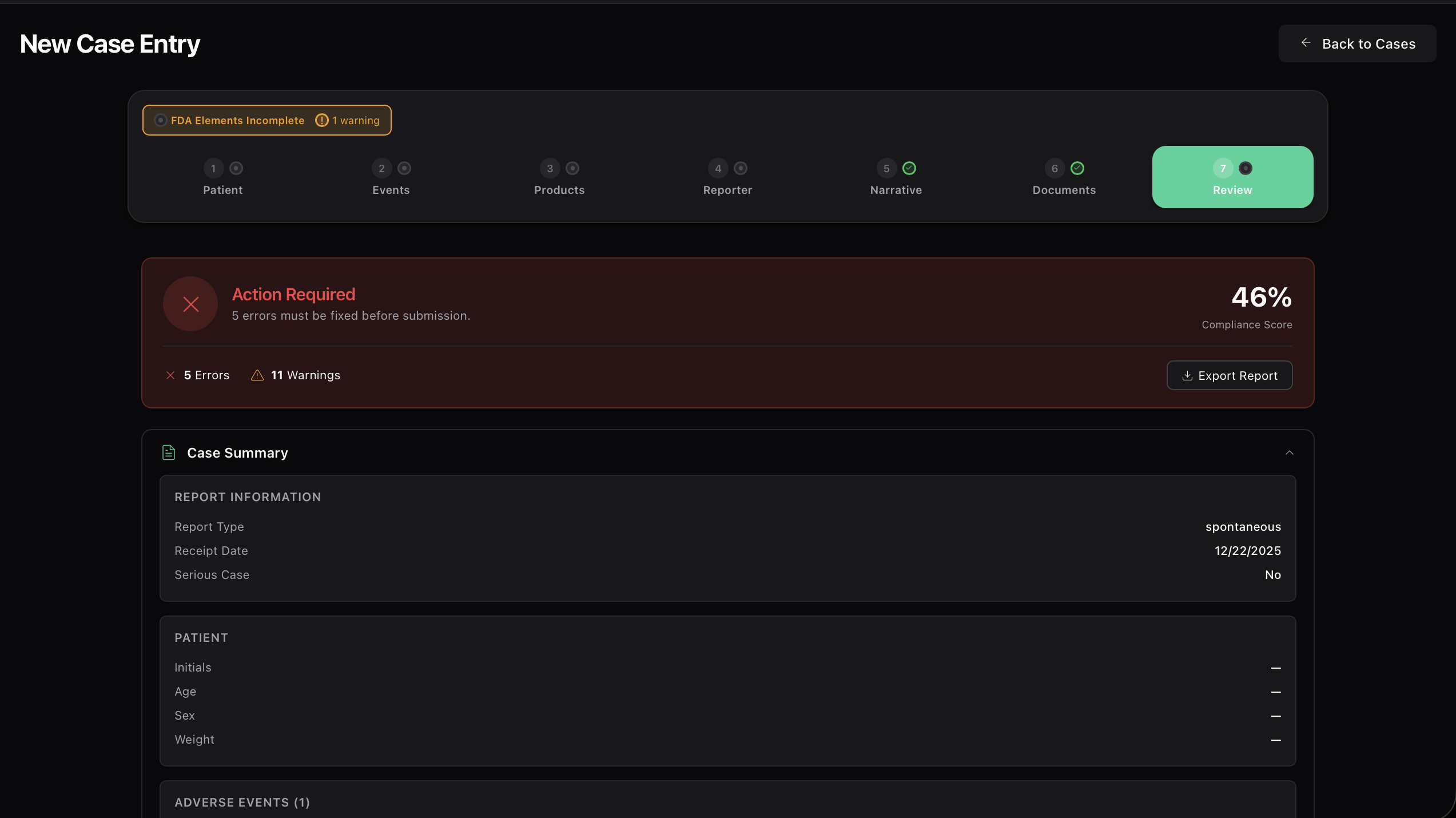
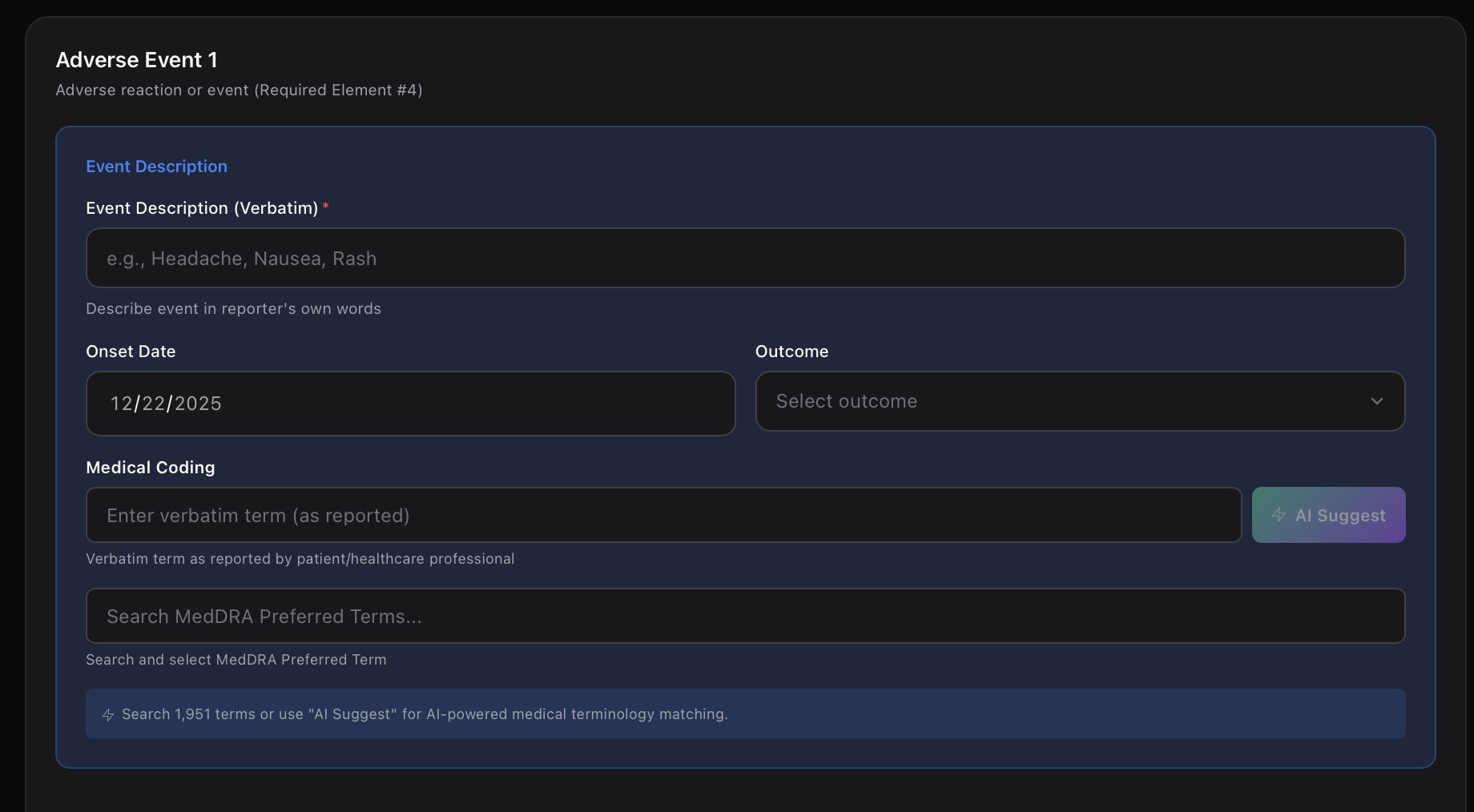
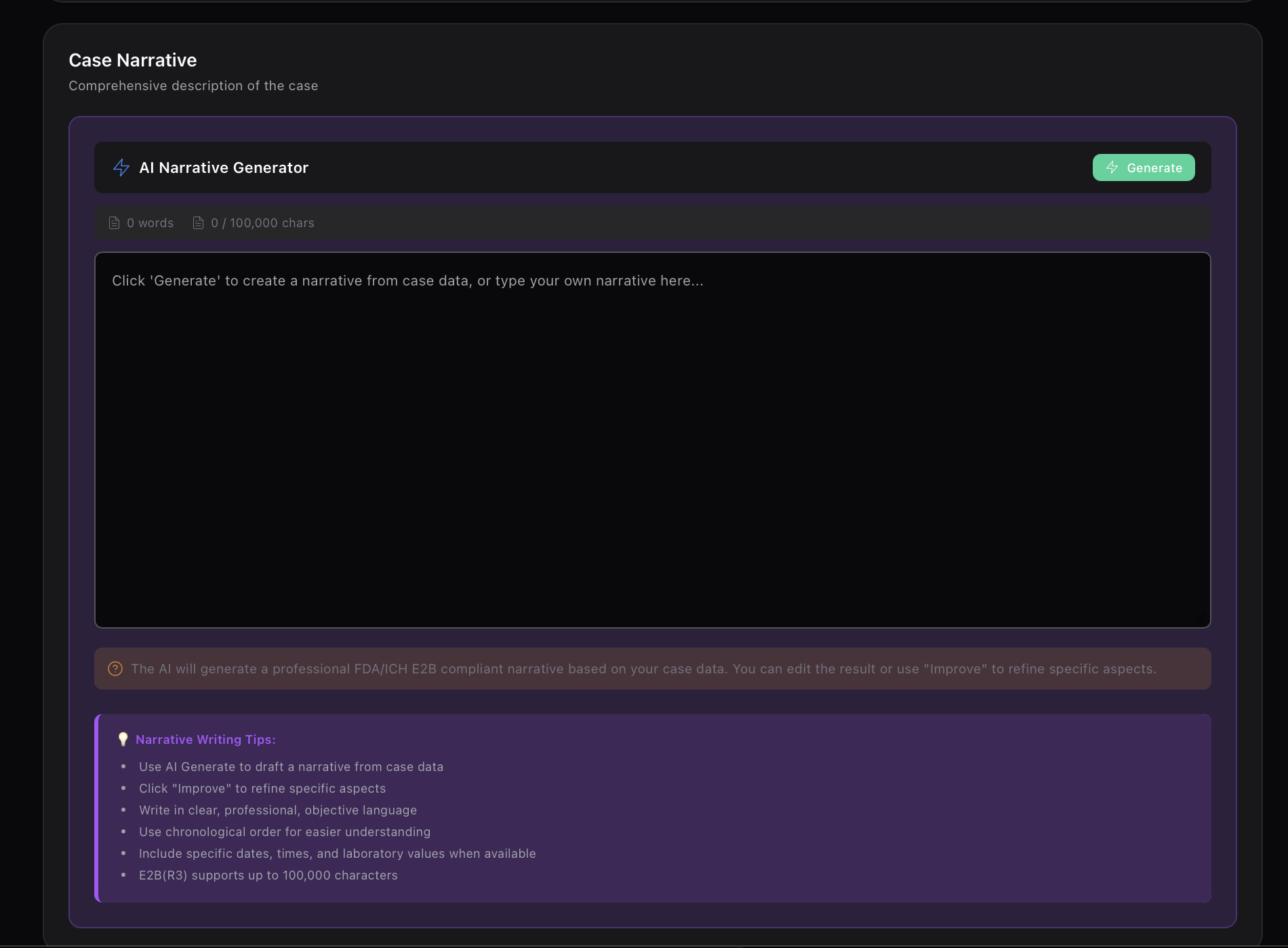
Automated safety signal detection. Identify risks before they become crises.
Start with Core PV. Add Intelligence when you're ready.
No implementation fees. No six-month deployments. Start today.
Core PV for early-stage teams
Scaling teams
Intelligence-powered
Global operations